Introduction
The soil in which the plant grows can be alkaline, neutral, or acidic. Most plants grow best in soil near neutral pH, but some plants prefer slightly acidic and others slightly alkaline soil. What is the soil pH in your garden? What happens to the pH of water that comes in contact with soil? All the answers are given in this simple project. Let’s BEGIN…
What is pH?
pH stands for potential of hydrogen which is a logarithmic scale used to specify the acidity or basicity of an aqueous solution. Solutions with a pH less than 7 are acidic while the solutions with a pH greater than 7 are basic. Pure water is neutral, which is neither acidic nor basic.
Soil pH
Soil pH is considered as a variable in soil which affects many chemical processes. In case of plant, it affect the plant nutrient availability by controlling the chemical forms of the different nutrients.
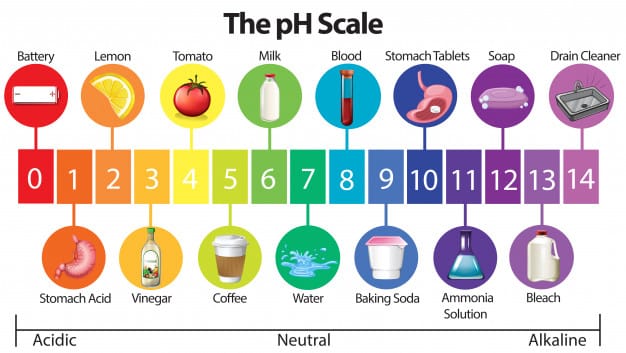
pH scale
It is the measure of degree of the acidity or basicity of a solution measured on a scale of 0 to 14, called pH scale. The midpoint on the pH scale represents neutral solution having pH 7 which is neither acid nor basic. pH value below 7.0 indicate acidity; numbers greater than 7.0 indicate basicity.
pH paper
pH papers are infused, saturated (mixture of indicators ) papers which are used to test for alkaline(or basicity) or acidity of the substance such as soil. pH papers are thin strips which change color when they come in contact with the material or solution being tested. 
Testing soil pH using pH paper
Below mentioned are the sequential steps to test the pH of a soil sample using the pH paper:
- Take a strip of pH paper.
- Gently dip the part of the paper into a soil. Wait for sometime and you will notice the color change in the pH paper strip.

- Compare the color obtained on the pH strip with pH chart which have a color-coded scale indicating the pH.

Output
The final output is shown in the figure. From the figure we infer that the soil pH is slightly basic in nature as the color on the pH scale is found out to be 8.

Conclusion
In conclusion, soil pH is an important factor to consider when growing plants. Knowing the pH of the soil can help you select the right plants for your garden. Testing the pH of the soil is easy and can be done with the help of pH papers. By following the steps mentioned in this lesson, you can easily find out the pH of the soil in your garden.