Introduction
Everybody knows that when you bring ice out of the fridge, it melts and turns into water after some time. And if you continuously heat water for some time, it starts boiling and turns into vapor. But what causes this change from solid to liquid and from liquid to gas? Come in: latent heat. Latent heat is the thermal energy absorbed or released by a substance during its phase change. But that is not all. Something really interesting happens when this heat changes the phase. To find out what it is, perform the experiment with us!
We’ll be using Dabble’s (our in-house developed mobile application that transforms your Smartphone into the numerous virtual shields) data logger while performing this experiment. You can download Dabble from Google Play.
Let’s get started!
Understanding Latent Heat
Before we begin, let’s briefly understand what latent heat is and why we are performing the given experiment.
What Is Latent Heat?
Latent heat is the thermal energy absorbed or released by a substance during its phase change without any change in temperature.
Latent Heat is also called Hidden Heat.
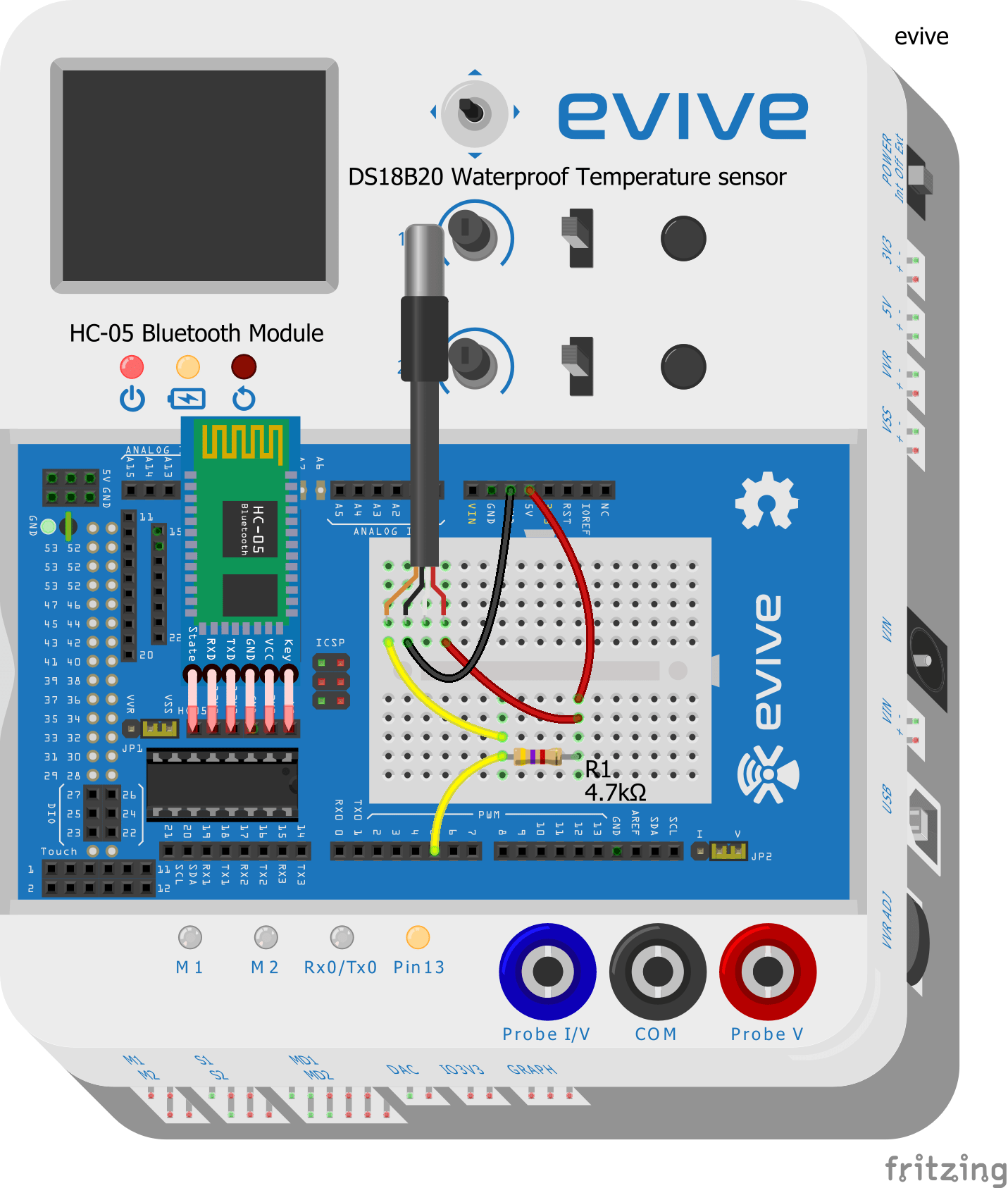
Connections
Let’s start by making the connections:
- Take the DS18B20 waterproof temperature sensor and connect its Signal pin to evive’s digital pin 5.
- Connect the VCC and GND pins of the waterproof temperature sensor to evive’s VCC and GND pins respectively.
- Connect a 4.7 kΩ resistor between the Signal pin and VCC of the temperature sensor.
- Connect the HC-05 Bluetooth module.
Make the connection as shown in the below figure: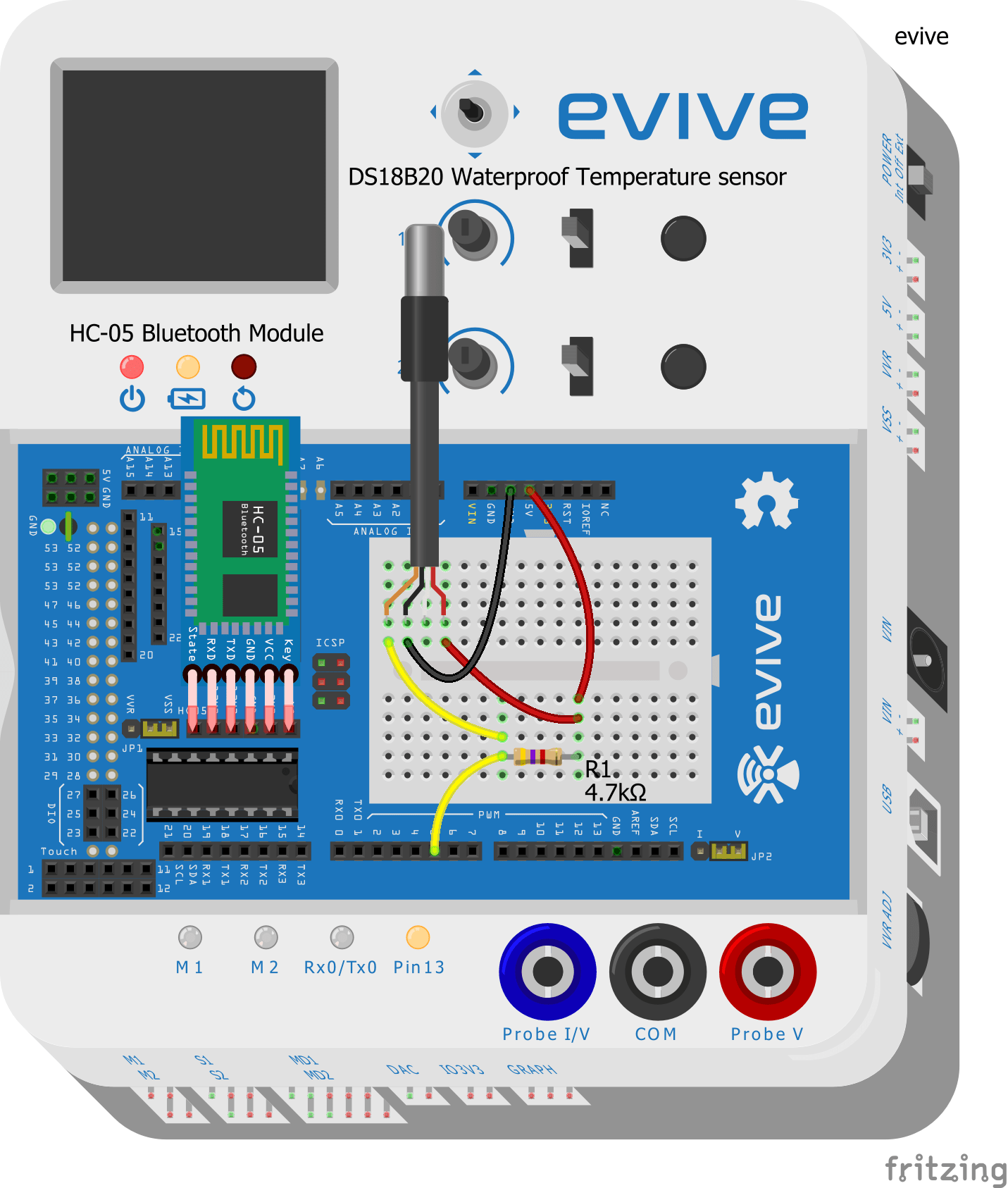
Setting Up the Experiment
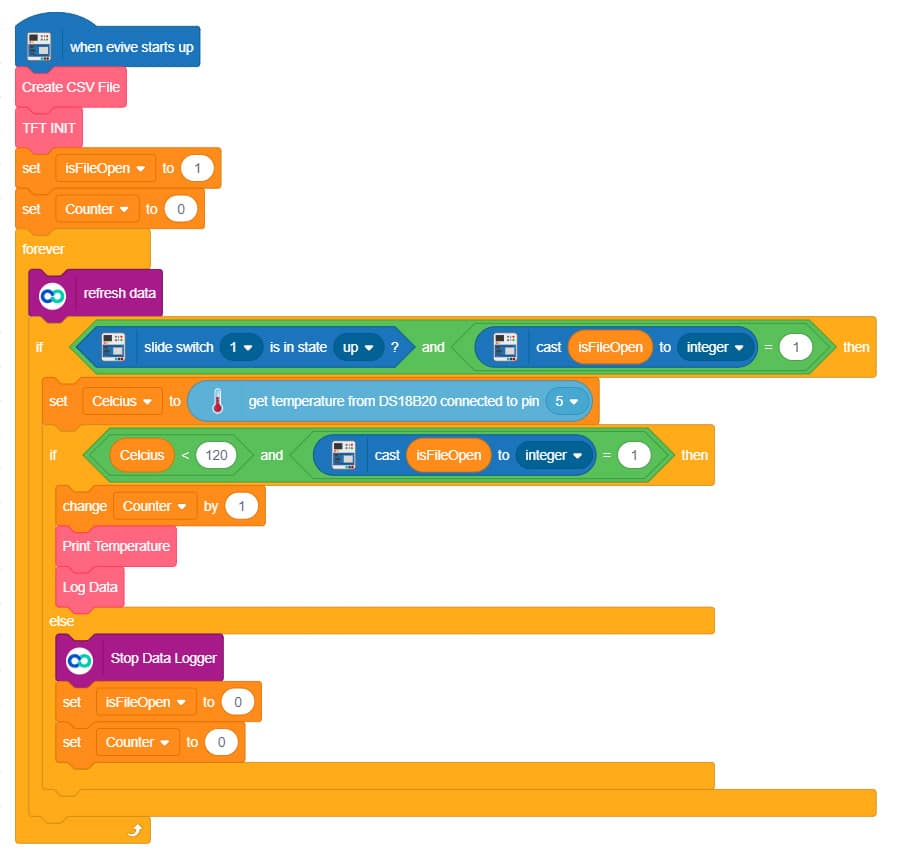
- Once done with the connections, turn on evive and make sure that you slide the Slideswitch1 downward.
- Now open Dabble on your Smartphone and select IoT module.
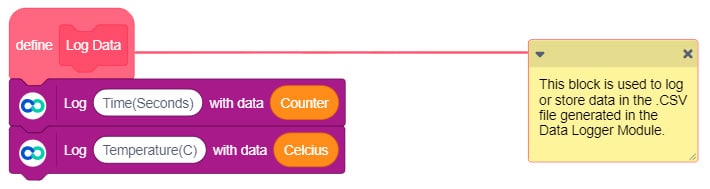
- In the IoT module, open the data logger. It stores data in real-time in a .CSV file which you can access later on.
- Now take an electric kettle or any other heating device and fill it with ice.
- Place the temperature sensor in the kettle.
- Now slide Slideswitch1 UP and turn the electric kettle ON.


If the temperature goes above 120°C then data logger stops logging data and then CSV file is created.
Code
PictoBlox is a graphical programming software based on Scratch 3.0 that makes learning how to code easy and fun. It allows you to interface and control evive with your computer and even uploads code to it. You can download PictoBlox from here.
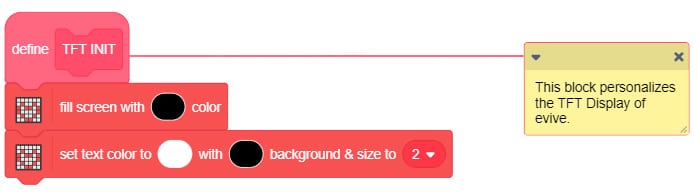
Upload the following PictoBlox (Scratch) Script to evive: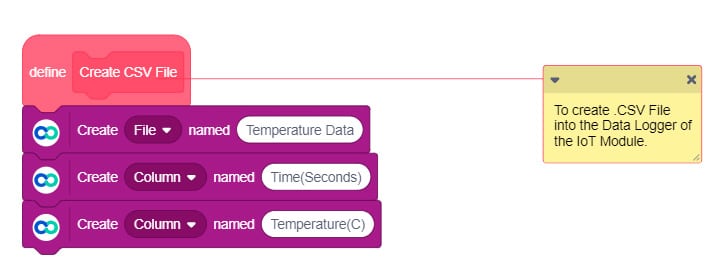

Observations
The data logger logs data in real-time. You will observe the following while performing the experiment:
- On heating the ice, the temperature initially increases which can be observed on evive’s TFT Display as well as on the screen of your Smartphone.
- Once the temperature reaches 0°C, the temperature no longer increases even though the energy is being supplied to the kettle until the ice melts completely.
- Once the ice completely melts and turns into the water, the temperature again starts increasing.
- Now the water starts boiling at 100°C the temperature again remains constant until the water is completely evaporated.
Explanation
Download the CSV File generated by the Data Logger and open it in your computer.
Plot the Temperature v/s Time graph using the data collected by the data logger. The graph look will look something like this: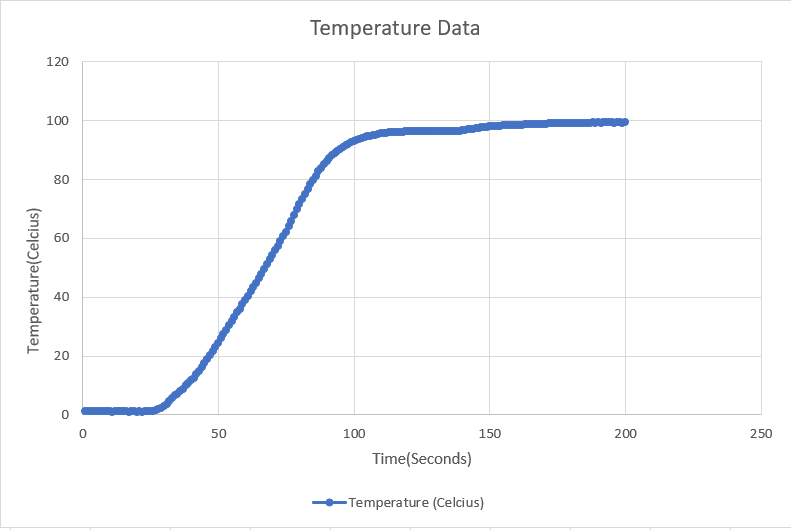
You will observe that the graph will be straight at two points:
- 0°C
- 100°C
When we start heating the ice, the molecules start vibrating leading to the increase the temperature. As soon as the temperature reaches 0°C, the molecules vibrate so much that the attractive force between them becomes weak and they start moving away. This continues till the ice is completely transformed from the solid state to liquid state i.e. water.
Throughout this process, all of the thermal energy is used to melt the ice. That is why the temperature remains constant.
This energy that is not seen as a temperature change is called Latent Heat. The energy that is used to change the phase from solid to liquid is called the Latent Heat of Fusion. And this point is said to be the melting of ice.
Once the ice completely turns into water, the temperature again starts increasing. As soon as the temperature reaches 100°C, the molecules again vibrate so much that the covalent bond between hydrogen and oxygen starts breaking, thus converting to vapors. All the energy given here is used to convert water to vapor which in turn keeps the temperature constant. This energy is called Latent Heat of Vaporization. And this point is said to be the boiling point of water.
Conclusion
With this, you are now familiar with the basics of latent heat. You can look up the internet more about it if you want. Happy tinkering!













![[CODEAVOUR 2021] project demonstration video 4-12 screenshot [CODEAVOUR 2021] project demonstration video 4-12 screenshot](https://ai.thestempedia.com/wp-content/uploads/elementor/thumbs/CODEAVOUR-2021-project-demonstration-video-4-12-screenshot-q7ynxvlv2t918md2w9xsnjiaauhor82z0jm2g1in34.png)